What are nanocoatings?
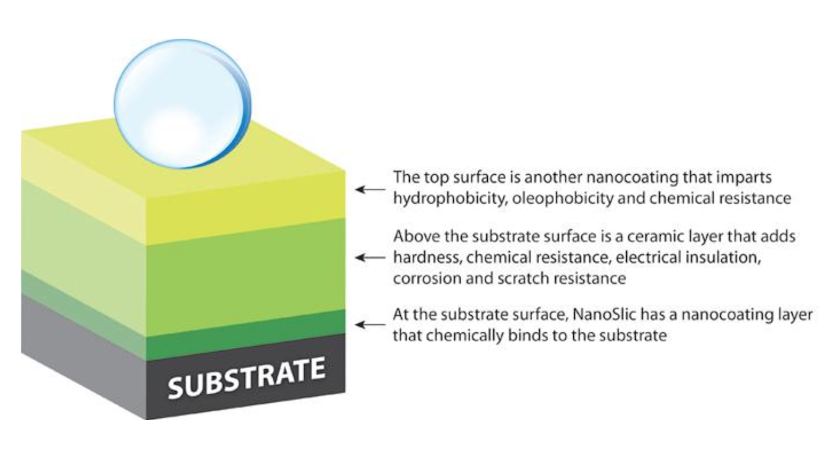
Nanocoatings are ultra-thin coatings or chemical structures that are built upon surfaces by a variety of methods. Technically, a nanocoating is a coating that's measured on the nanoscale. To compare, the typical automotive paint is approximately 125 microns or 125,000 nanometers. At the nanoscale, quantum physics comes into play. Nanocoatings can be structured one molecule thick, or they can be built up from multiple molecular layers. Then there are hybrid nanocoatings, such as NanoSlic, which combine multiple layers of nanocoatings to deliver a wide range of benefits.
By 2025, it is estimated that the global market value for nanocoatings will climb to $17.2 billion, which is an all-time high. The nanocoatings market is all-encompassing, advancing diverse applications, including anti-fouling, fingerprint resistance, water repellency for clothing, and even bacteria growth prevention to curb the infectious spread.
But the growth of protective coatings for electronics is unparalleled, as billions of electronic devices deploy into diverse and challenging environments. These devices require protection against threats, including rain, sweat, submersion, pollution, and more. As conventional protection methods can fall short for these new, miniaturized, edge electronics, protective nanocoatings offer promising safeguarding solutions and massive growth potential.
Protective Nanocoatings for Electronics
Years ago, desktop computers were safely perched in offices. Today, smaller devices must operate with flexibility, independent of environments. For connected components, familiar elements have become failure-inducing variables. Humidity, corrosives, contaminants, liquids, temperature extremes, and electrical motion can all wreak havoc. Sensitive circuitry requires protection. For years, the paint and coatings industry has used conventional means to meet this need. Barrier, inhibitive, sacrificial, and combination coatings,
Unfortunately, these coatings can be too thick, too porous, and too bulky for miniaturized devices. Meanwhile, an increasing emphasis on sustainability is brewing in the industry.
Nonbiodegradable, toxic chemicals will soon no longer be workable options for component protection. Foreseeing these roadblocks, manufacturers are shifting strategies, breaking with traditional coatings. Protective nanocoatings, thinner than a piece of paper, offer a robust alternative.
Why Are Protective Nanocoatings a Better Solution?
Like traditional coatings, nanocoatings and thin-film alternatives are deposited on a surface to improve protection. The difference is that a protective nanocoating can be as thin as a few nanometers, typically starting at 300 nm. This profile minimizes unit weight and mass, which proves useful for compact devices. Though super thin, their protection from ice, wear, pollutants, heat, and corrosion surpass traditional coatings. Alternative benefits can include suitable dielectric and thermal management properties and hydrophobic and oleophobic characteristics. The protection can be tailored to circumstances by selecting and combining various materials. Nanocoatings and thin films are functionalized to solve specific business challenges; an efficient, competitive solution. Increasing global regulations aren't typically an issue with nanocoatings, as most are REACH and RoHS compliant with minimal to no VOC content. Many protective nanocoating vendors practice proactive chemical management, material oversight, and corporate sustainability. These focuses, coupled with green material usage and preparation for repair and recycling, make for a sustainable solution. Market Growth Rate and Key Growth Drivers Analysts expect the global nanocoatings market to grow at a CAGR of 17.22% from 2018 to 2025. Growth drivers include the ability to engineer films for:
• Liquid splash and immersion protection
• Corrosion resistance
• UV resistance
• Solvent and chemical protection
• Transparency
• Durability
• Advanced bonding between surface and coating
• Capacity to increase the lifespan and durability of the substrate
• Flame resistance and fuel and gas barriers
• Reduced manufacturing costs
• Replacement of toxic inhibitors with minimal VOC emissions
Protective Nanocoatings Applications Consumer Electronics
The consumer electronics market size valued at USD 1 trillion in 2019 is estimated to grow at a CAGR of over 7% between 2020 and 2026. Cell phone damage alone due to liquid exposure is estimated to cost the industry upwards of $97.1B annually.
Tired of repairs and replacements, waterproofing is table stakes for success with consumers. Waterproofing is only one need for consumer devices. Hearables and wearables, like smartwatches and earbuds, experience adverse operating conditions. Sweat, steam, perfume, and cleaning fluids are but a few common hazards that jeopardize circuitry. Smart home security products (cameras, video doorbells, alarms, and smart locks), predicted to become a $4.3 billion industry by 2023, is another product line in need of protection. Pollution, moisture, corrosives, and chemicals can cause unexpected or immediate failure.
Protective nanocoatings can exceed the industry standards for protection, including the IPC IP ratings, at an economical price. This solution is a competitive candidate for consumer electronics protection. Medical Devices The medical device industry is exponentially expanding, with growth fostered by an aging population and a desire to manage health proactively. By 2025, the medical device industry will be valued at $612.7 billion worldwide. Currently, one in 11 adults around the globe lives with diabetes. While glucose meters and insulin pumps are helpful devices, they are still vulnerable to splashes and spills. Diagnostic imaging also comprises an impressive market, valued at $40 billion. EEG analyzers face cleaning fluids, body fluids, and other liquids daily, requiring protection. Finally, over 900 million people will experience hearing loss by 2050. This population will subject their hearing aids to sweat, moisture, humidity, and body oils. Yet, hearing aid components can be as small as a pencil eraser. The healthcare community is actively exploring the robust, low-mass protection of nanocoatings. Industrial In the next four years, the global market size for the Industrial Internet of Things (IIoT) will reach $110.6 billion. This market growth necessitates protection from common industrial failure conditions. These environments include corrosives, contaminants, inclement weather, sulfuric gases, and petroleum products. In the distributed energy generation (DEG) global market, expected to reach $573.7 billion by 2025, similar concerns exist. Digital logic controllers and other DEG products need to be resilient to avoid downtime and failure rates.
Meanwhile, the IoT market in oil and gas will hit $39.40 billion globally by 2023. To implement connected OT edge in these environments, manufacturers must put reliability first. Protective nanocoatings and thin-film solutions, with excellent corrosion resistance properties and beneficial thermal and electrical properties, compare well to existing solutions that use operator-dependent processes that are not as repeatable. Automotive The automotive industry is making a paradigm shift towards autonomous products and technologies. By 2030, electronics will account for 50% of a car's total cost worldwide, creating profitable opportunities. Global revenue for the connected car market will reach $166 billion by 2025. Autonomous vehicles (AV) are at the forefront of change, expected to comprise a quarter of the marketplace by 2040. AVs mandate unprecedented reliability; occupant safety is a paramount concern. Achieving this type of resiliency may seem impossible, given the harsh service environment. Oil, gas, antifreeze, corrosives, temperature extremes, contaminants, pollutants, and humidity are all obstacles.
The need for low-weight, low-bulk, sustainable reliability makes protective nanocoatings a promising option. IoT Forecasts suggest that by 2030, around 50 billion IoT devices will be in use around the world, creating a massive web of interconnected devices spanning everything from smartphones to kitchen appliances. There will be 53.63 million active smart city connections in the European Union alone in the next couple of years. Cameras monitor traffic, while noise sensors alert personnel to the potential of danger. Smart cities must run dependably with cost contained and constituents safe. That means every component in the network must reliably run too. Everything from utility meters and cell phone towers is mission-critical, but salt fog, humidity, snow, and dust storms are genuine threats to these IoT components. Sensors, switches, and hubs are also devices that need thin, lightweight protection.
Scalability, sustainability, and minimal bulk are enticing benefits for protective nanocoatings in the IoT arena. The Future of Protective Nanocoatings The protective nanocoatings industry is well-established, but some measures need to be adopted to adopt the new technology further to encourage its capable growth. More time and financial investment are required. Technological research is necessary. And it must fall in line with governments, research councils, and regional development agencies.
Further development of expertise must occur, and stakeholders must identify gaps in value chains. Stakeholders must use networking opportunities and open access facilities to develop new technology. Most importantly, future products need to be produced responsibly. Public perception and risks must be addressed as products and processes mature. Any potential for the environment, health, life cycle analysis, and safety risks must be minimized if protective nanocoatings are to be an acceptable mainstream solution. The Big Picture Independent of industry, the protective nanocoatings marketplace growth potential is considerable, as protecting electronics becomes increasingly challenging as devices shrink. Beyond protection, weight, and mass, dielectric and thermal management also need to be considered a protective solution. Based on advancing regulations, environmentally sound solutions such as nanocoatings are gaining popularity. Ultimately, delivering resilience and durability helps minimize costs associated with repairs, warranty claims, and service calls. Reliability mitigates liability, improving uptime, and drives incremental product value. As the market continues to evolve, the future for protective nanocoatings looks bright. Thin films and nanocoatings' custom protection is an enticing solution over traditional protection across industries and offerings, ensuring that market value and interest in the technology will continue to trend upwards.
Protection of infrastructure from corrosion is essential in preventing costly failures, with risk to human life and its structure. This is frequently achieved through the use of complex, multi-layered coating systems. Current organic coating systems designed for harsh environments comprise several different coating layers, each providing a different set of properties. A basic system usually consists of three layers, including a primer coat, an intermediate coat, and a final topcoat. Primer coats are typified by epoxy-based formulations containing a relatively high loading of a more anodic metal such as zinc (zinc-rich), which provides sacrificial protection to the metal substrate. Intermediate coats are usually formulated around a solvent/epoxy base with a pigment blend containing a relatively large fraction of micaceous iron oxide. The intermediate coat or tie coat serves to promote adhesion between the primer coat and the topcoat layers. The intermediate coat may also provide barrier-type protection against corrosive species such as water, ions, or oxygen, slowing their diffusion to and from the metal surface. However, it is recognized that there are some limitations to these barrier properties due to the permeability of organic coatings to such corrosive species. Finally, the topcoat of the system is normally composed of polyurethane or polysiloxane. These materials usually offer UV resistance in addition to any aesthetic finish. A key component of these systems in high-risk environments with a significant corrosion risk is the zinc-rich primer. These primers have been widely used since the 1930s for the protection of steel structures. Unlike regular organic paints, they provide galvanic protection at edges or the places where the coating is defective. In the FHWA-sponsored "PACE" study, one of the coatings with zinc-rich primers performed best compared to other generic types of coatings evaluated under similar conditions. Zinc-rich coatings are described in several industry and military specifications. SSPC Paint 202 is an industry specification that categorizes zinc primers according to vehicle types. Type II coatings are the most common, involving organic vehicles such as epoxies and moisture-cured urethanes. The protection of steel by a zinc-rich coating is achieved mainly via two effects: the barrier Improvements in Anti-Cor By Integrating Graphene Nano-Platelets into C By Matthew Sharp, William Weaver, Lynn Chikosha and Sam Whitehead, Applied Graphene Materials, Redcar, UK PAINT & COATINGS INDUSTRY • 25 Corrosion Performance o Coating Systems effect imparted by the coating itself and galvanic action. The zinc particles in the coating provide galvanic protection. Three conditions are essential for the galvanic process to occur on steel:
1. Zinc particles must be in electrical contact with each other.
2. Zinc particles must be in electrical contact with the steel.
3. A continuous electrolyte must exist between the zinc particles and the steel.
The first two conditions are met by zinc-rich coatings when containing a sufficiently high zinc content. The third condition is fulfilled when a steel panel bearing a zinc-rich coating is wetted by an electrolyte film such as a salt solution. The protective action of zinc-rich coatings may be broken down into two stages. The first stage is a relatively short period in which zinc particles' galvanic protection of the steel occurs. After this period in which the zinc is consumed, the galvanic action between the steel and zinc gradually disappears. The second stage is long-term barrier protection that is attributed to greater resistance of the coating to the permeation of aggressive species such as water, oxygen, and salts because the pores in the coating are blocked by the zinc corrosion products and inhibition of the steel surface by the zinc corrosion products. The properties of graphene, a single layer of graphite, have been talked about for many years. High mechanical and electrical properties have a high aspect ratio, endowing the ability to function as a barrier additive. When incorporated into an organic coating system or host matrix, graphene nanoplatelets (GNPs) provide a highly tortuous pathway that impedes corrosive species' movement towards the metal surface, three a passive corrosion protection mechanism.
Also, very small additions of GNPs decrease water vapor transmission rates,4 indicating a barrier-type property, while some authors also suggest an electrochemical activity provided by graphene within coatings.5 Formulating a High-Risk-Environment System Using Graphene The use of graphene nanoplatelets combined with zinc has been proposed and shown to have both positive and negative impacts, depending on loading6 when incorporated directly in the zinc-rich primer.

.png)




No comments:
Post a Comment